-
 Solved If pH the methyl red solution is changed 4, Chegg.com
Solved If pH the methyl red solution is changed 4, Chegg.com
-
 Acids, Indicators and Buffers
Acids, Indicators and Buffers
-
4. Cakculate the pKa of methyl from following | Chegg.com
-
 Methyl red has the following structure: undergoes a color from red to yellow as a solution gets more basic. Calculate an approximate pH range for which methyl red is useful.
Methyl red has the following structure: undergoes a color from red to yellow as a solution gets more basic. Calculate an approximate pH range for which methyl red is useful.
-
 VOLUMETRIC ANALYSIS Volumetric analysis is an analysis in which the amount of the unknown is calculated from a known volume of added solution. ppt video online download
VOLUMETRIC ANALYSIS Volumetric analysis is an analysis in which the amount of the unknown is calculated from a known volume of added solution. ppt video online download
-
 Answered: Methyl has a pk, of 5.0 and is | bartleby
Answered: Methyl has a pk, of 5.0 and is | bartleby
-
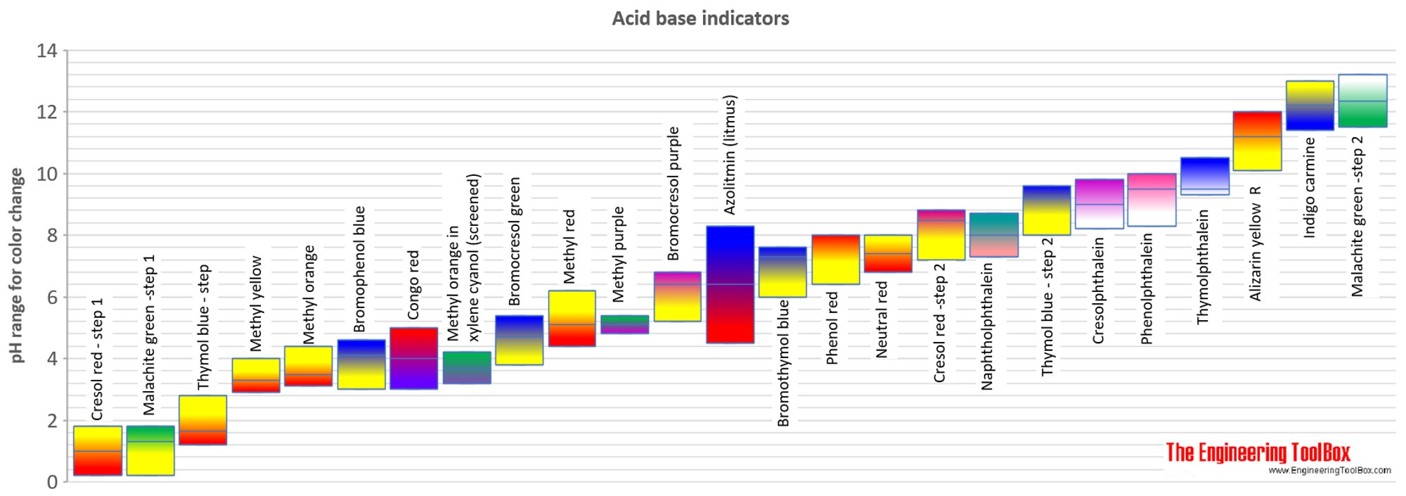 Acid and Base pH
Acid and Base pH
-
pKa of Red Purpose: The pKa ' of red will be determined by measuring absorbance spectra as a function pH.
-
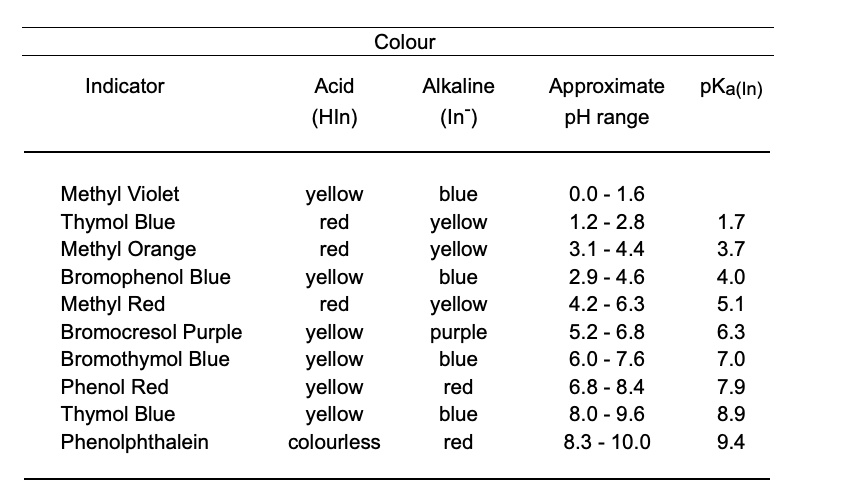 SOLVED: Acid (HIn) Alkaline (In" ) Approximate pH range pKa(ln) Methyl Violet Thymol Blue Orange Bromophenol Blue Methyl Red Bromocresol Purple Bromothymol Blue Phenol Red Thymol Blue Phenolphthalein yellow
SOLVED: Acid (HIn) Alkaline (In" ) Approximate pH range pKa(ln) Methyl Violet Thymol Blue Orange Bromophenol Blue Methyl Red Bromocresol Purple Bromothymol Blue Phenol Red Thymol Blue Phenolphthalein yellow
-
 Natural Plant Acid-Base Indicator Determination of Their pKa Value
Natural Plant Acid-Base Indicator Determination of Their pKa Value
-
 Structures diazo pH indicators. (A) Main of methyl | Download Scientific Diagram
Structures diazo pH indicators. (A) Main of methyl | Download Scientific Diagram
-
 Structures diazo pH indicators. (A) Main of methyl | Download Scientific Diagram
Structures diazo pH indicators. (A) Main of methyl | Download Scientific Diagram
-
 Methyl -
Methyl -
-
 6 Determination of pKa of red - Studypool
6 Determination of pKa of red - Studypool
-
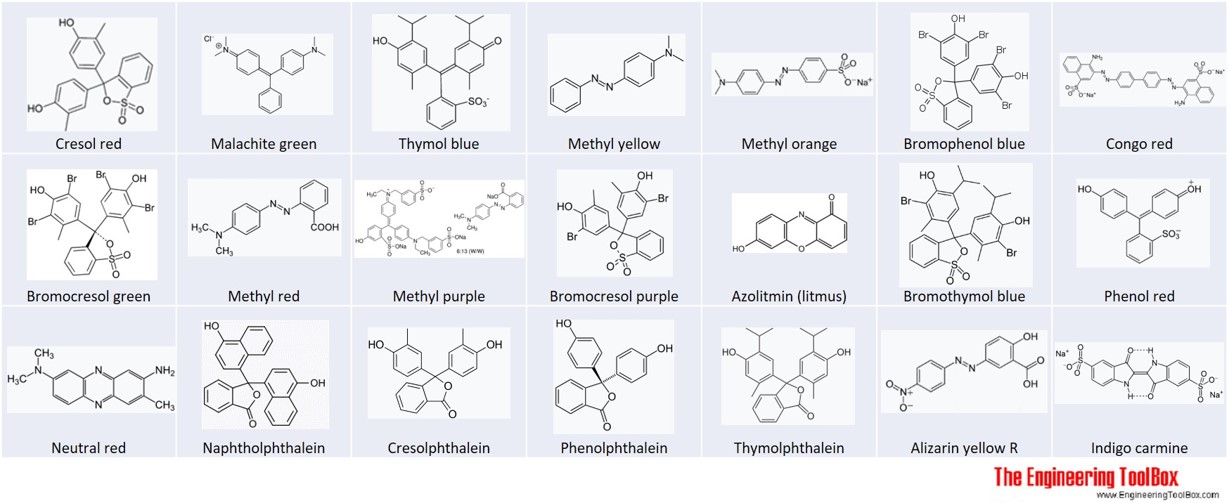 Acid and Base pH
Acid and Base pH
-
 Solved Methyl red is good choice as an indicator for the | Chegg.com
Solved Methyl red is good choice as an indicator for the | Chegg.com
-
 Solved Calculate the K, and methyl red from the | Chegg.com
Solved Calculate the K, and methyl red from the | Chegg.com
-
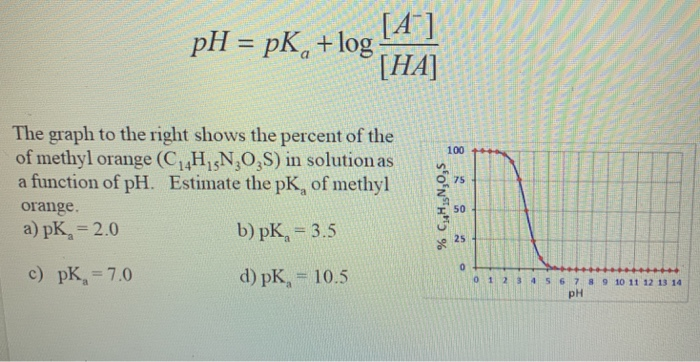 Solved ООН pk, = 4.8 pK,-5.1 Methyl has acidic and basic | Chegg.com
Solved ООН pk, = 4.8 pK,-5.1 Methyl has acidic and basic | Chegg.com
-
 SOLVED:Methyl red has a pKa of 5.0 and is red its acid form and yellow in its basic form. If several drops of this indicator are placed in 25.0
SOLVED:Methyl red has a pKa of 5.0 and is red its acid form and yellow in its basic form. If several drops of this indicator are placed in 25.0
-
 EXPERIMENT 14. ACID DISSOCIATION OF METHYL
EXPERIMENT 14. ACID DISSOCIATION OF METHYL
-
 Chem435. Physical Chemistry Laboratory. Acid Dissociation Constants.
Chem435. Physical Chemistry Laboratory. Acid Dissociation Constants.
-
 A Simplified Method for Finding pKa of Acid–Base Indicator
A Simplified Method for Finding pKa of Acid–Base Indicator
-
 Finding pKa of pH indicator Methyl red - chemistry
Finding pKa of pH indicator Methyl red - chemistry